Seminari de Biofísica: Monolayer model membranes, a plethora of methods to tackle interesting biological systems
Data: 5 d’octubre de 2017, a les 15h
Lloc: Sala de Conferències, Edifici TR1, ESEIAAT, Universitat Politècnica de Catalunya. C/Colom 1, Terrassa
Organitza: Secció de Biofísica de la Societat Catalana de Biologia
Conferenciant: Christian Salesse, Université Laval et Centre de Recherche du CHU de Québec-Université Laval
Idioma: Anglès
RESUM
Cell membranes are made of a complex mixture of lipids and proteins. These membranes include less than 100 different types of lipids when taking into account the different types of acyl groups. In addition, one can find integral membrane proteins, such as the G-coupled receptors, as well as membrane-attached proteins through N- and/or C-terminal transmembrane segments or amphipathic segments. Proteins can also be membrane-anchored through different types of acylations.
Model membranes are thus necessary to decrease this level of complexity and decipher specific interactions taking place between proteins and lipids.
“The monolayer model membrane is very well suited to study the preferential binding of peripheral proteins to lipids”
It has also the advantage to allow studying protein binding to lipids found on the inner or the outer leaflet of membranes. Moreover, a large number of powerful methods have been developed to study this type of model 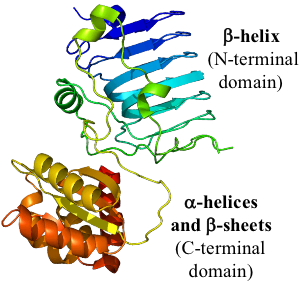 membranes such as X-ray and neutron reflectivity, absorption, fluorescence, and infrared spectroscopy, fluorescence and Brewster-angle microscopy and other physical methods (ellipsometry, SHG, etc).
membranes such as X-ray and neutron reflectivity, absorption, fluorescence, and infrared spectroscopy, fluorescence and Brewster-angle microscopy and other physical methods (ellipsometry, SHG, etc).
“During this seminar, I will present data using different methods to gather information on protein binding to lipid monolayers as well as the characterization of integral membrane proteins spread as monolayers”
The examples will include the binding of:
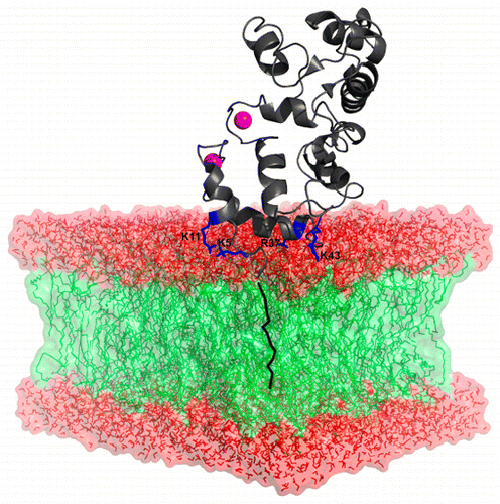
1) Recoverin, a protein whose membrane binding is controlled by a mechanism of calcium-myristoyl switch (see the conformational change taking place upon calcium binding in the opposite figure)
2) Retinitis pigmentosa 2 (RP2), a protein whose structure includes a hydrophobic beta helix (see opposite figure)
3) Rhodopsin, a visual pigment
4) Bacteriorhodopsin, a protein from Halobacteria acting as a proton pump.
 I will show that calcium-bound recoverin undergoes a preferential binding to negatively charged lipids as a result of the extrusion of its myristoyl group and positively charged amino acids (K11, K5, K37 and R43, see upper figure). In contrast, RP2 preferentially binds lipids bearing saturated fatty acyl chains. Moreover, the secondary structure of rhodopsin was found to be unaltered when quickly spread in monolayers at medium values of initial surface pressure. Finally, bacteriorhodopsin is showing a very particular signature in infrared spectroscopy which allowed characterization of its properties.
I will show that calcium-bound recoverin undergoes a preferential binding to negatively charged lipids as a result of the extrusion of its myristoyl group and positively charged amino acids (K11, K5, K37 and R43, see upper figure). In contrast, RP2 preferentially binds lipids bearing saturated fatty acyl chains. Moreover, the secondary structure of rhodopsin was found to be unaltered when quickly spread in monolayers at medium values of initial surface pressure. Finally, bacteriorhodopsin is showing a very particular signature in infrared spectroscopy which allowed characterization of its properties.